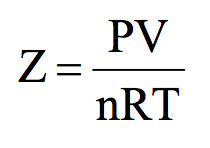
Share
A gas which obeys the ideal gas equation, PV = nRT under all conditions of temperature and pressure is called an ideal gas. There is no gas which obeys the ideal gas equation under all conditions of temperature and pressure. The gases are found to obey the gas laws if the pressure is low or […]
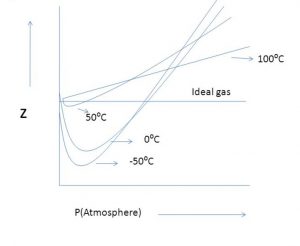
Real Gases - Chemistry, Class 11, States of Matter
Real Gases - Chemistry, Class 11, States of Matter
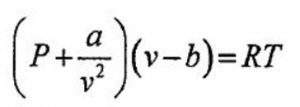
Real Gases - Chemistry, Class 11, States of Matter
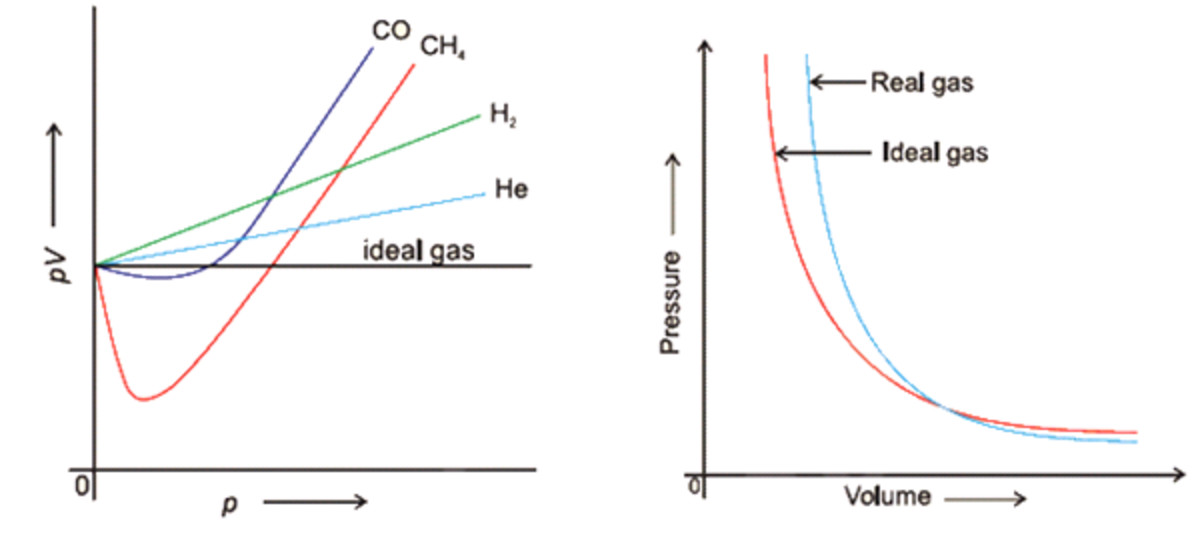
Real Gases - Chemistry, Class 11, States of Matter
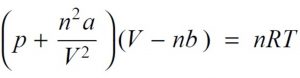
Real Gases - Chemistry, Class 11, States of Matter
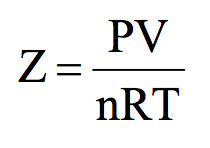
Real Gases - Chemistry, Class 11, States of Matter
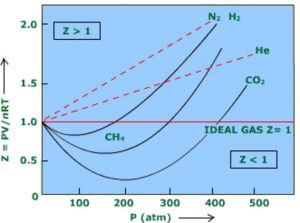
Real Gases - Chemistry, Class 11, States of Matter
Related products

Gas Compressibility Factor Calculator Excel SpreadsheetLow Cost Easy to Use Spreadsheets for Engineering Calculations Available at Engineering Excel Spreadsheets
Solved] Why is the compressibility factor less than 1 at most conditions?

Which of the following statements is/are correct? (a) all real gases are less compressible
Slope of graph of compressibility factor(Z) with pressure(P) for
You may also like

The Best Sustainable Eco Friendly Women's Shorts

Castor Oil Pack Wrap for Breast -9.6 Inches Reusable Organic Cotton Castor Oil Compress Pads Less Mess for Fibrocystic Breasts (D/E/F) : Health & Household

Small Baby Underwear, Unisex, 3-Pack

Fit Muscle Manmen's Slimming Body Shaper Tank Top - Nylon Compression Vest For Gym & Workout