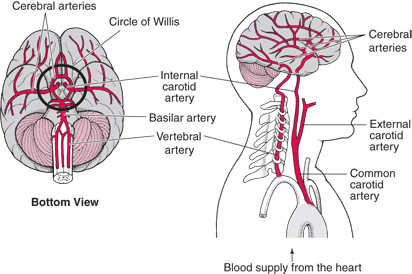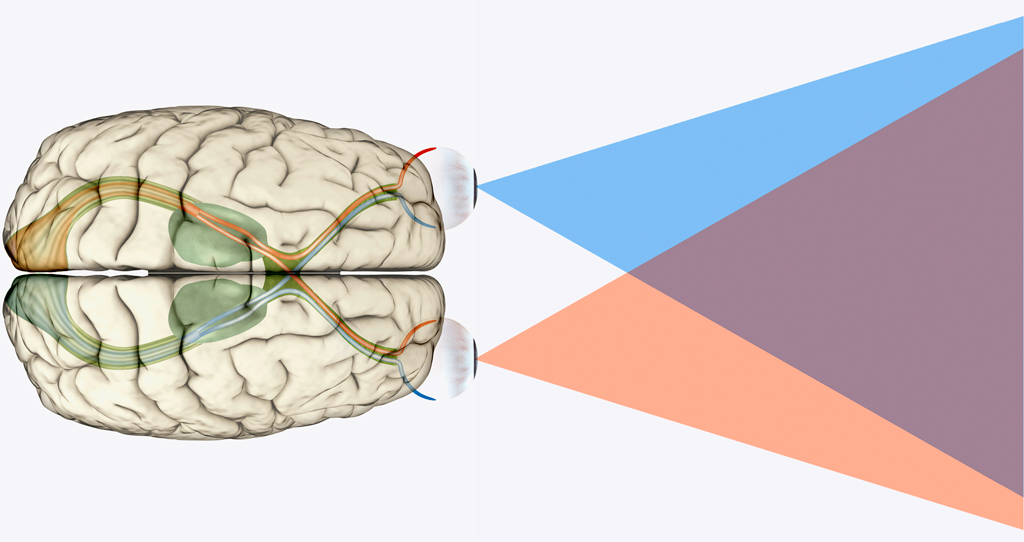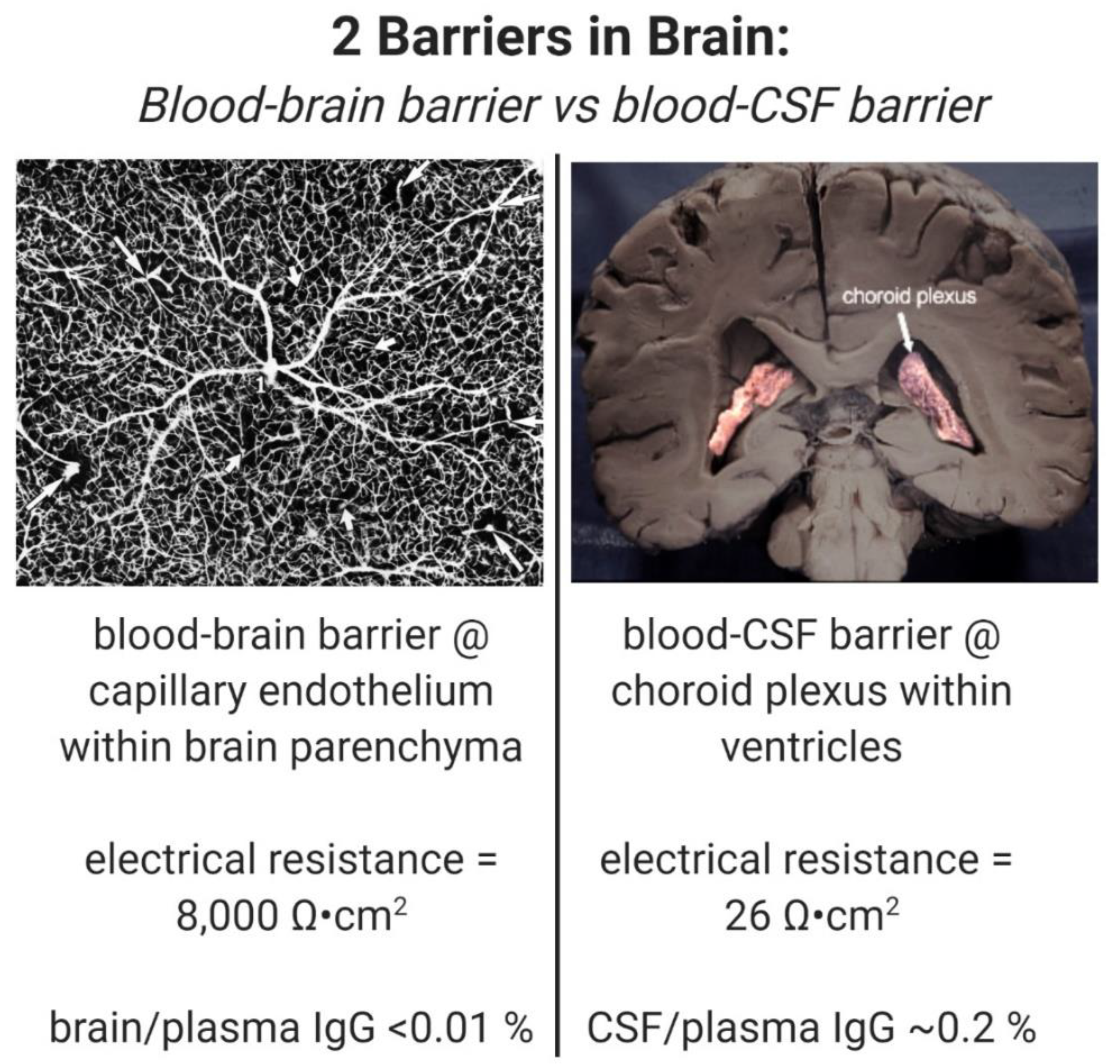
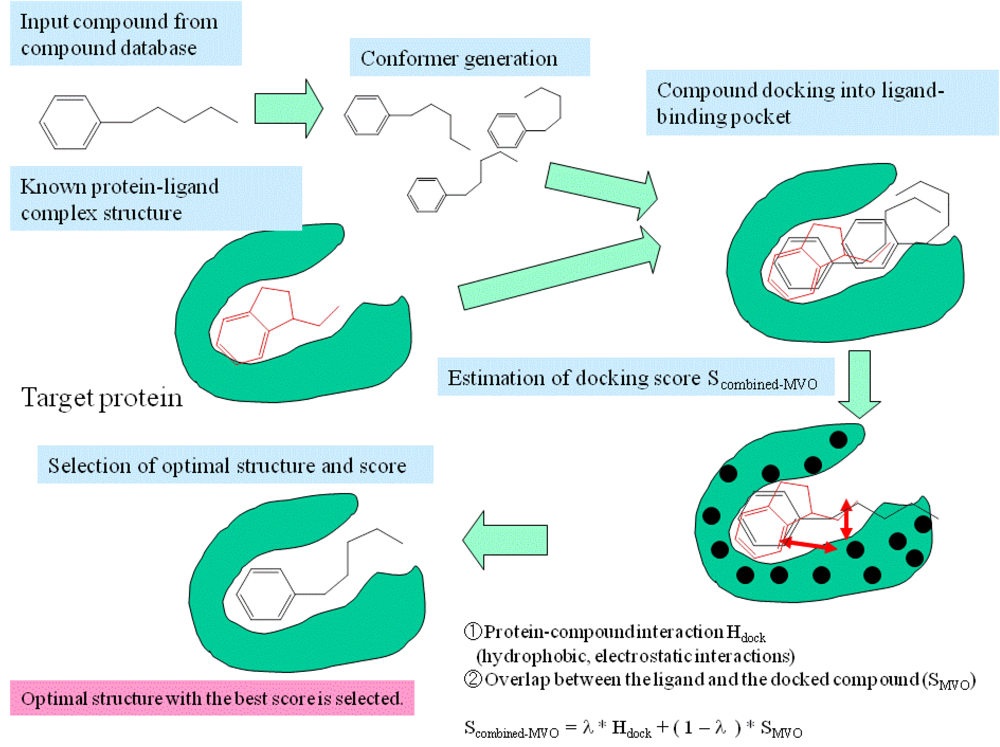
Despite the enormity of the societal and health burdens caused by Alzheimer’s disease (AD), there have been no FDA approvals for new therapeutics for AD since 2003. This profound lack of progress in treatment of AD is due to dual problems, both related to the blood–brain barrier (BBB). First, 98% of small molecule drugs do not cross the BBB, and ~100% of biologic drugs do not cross the BBB, so BBB drug delivery technology is needed in AD drug development. Second, the pharmaceutical industry has not developed BBB drug delivery technology, which would enable industry to invent new therapeutics for AD that actually penetrate into brain parenchyma from blood. In 2020, less than 1% of all AD drug development projects use a BBB drug delivery technology. The pathogenesis of AD involves chronic neuro-inflammation, the progressive deposition of insoluble amyloid-beta or tau aggregates, and neural degeneration. New drugs that both attack these multiple sites in AD, and that have been coupled with BBB drug delivery technology, can lead to new and effective treatments of this serious disorder.
Pharmaceuticals, Free Full-Text

NeilMed Pharmaceuticals - Sinus Rinse, All Natural Relief - 100 Premixed Packets

Pharmaceuticals Free Full-Text Preclinical Efficacy And, 54% OFF

Online Medical Store: Order Medicine Online & Get Fastest Delivery

CADILA Pharmaceuticals Skin Shine Rich Skin Brightening & Lightening Cream Pack of 4 - Price in India, Buy CADILA Pharmaceuticals Skin Shine Rich Skin Brightening & Lightening Cream Pack of 4 Online

Dermarest Eczema Medicated Lotion, Fragrance Free 4 Fl Oz / 118 Ml (Pack of 4) : Health & Household

m.media-/images/I/81QNwMH+VAL._AC_UF1000
Lack of effect of pantoprazole on the pharmacodynamics and pharmacokinetics of warfarin. - Abstract - Europe PMC

Generic drug - Wikipedia
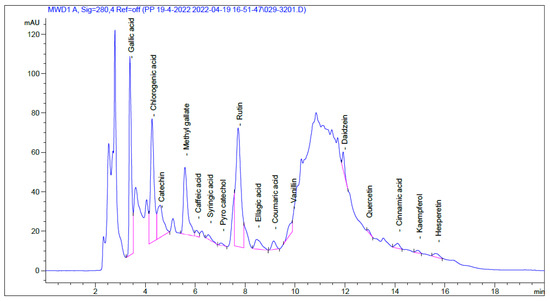
Pharmaceuticals, Free Full-Text
)
Glenmark Pharma launches first FDC drug Indamet for asthma patients
TCI offers free birth control in Tulsa County

Pharmaceuticals, Free Full-Text