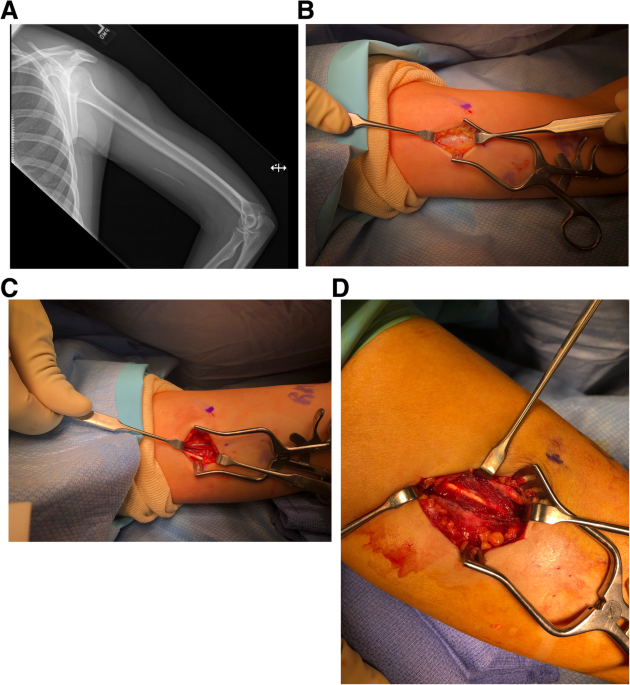
Background Subdermal etonogestrel implants are highly effective contraceptive methods. Despite standardization of insertion technique by the manufacturer, some implants are inadvertently placed too deeply within or below the plane of the biceps brachii fascia. Placement of these implants in a deep tissue plane results in more difficult removal, which is not always possible in the office setting. In rare cases, surgical removal by an upper extremity surgeon is warranted. Case presentation Here we present 6 cases of etonogestrel implants located in a subfascial plane requiring removal by an upper extremity surgeon. Implants were all localized with plain radiography and ultrasound prior to surgical removal. All cases had implants located in the subfascial plane and one was identified intramuscularly. The average age was 28 years (19–33) and BMI was 24.0 kg/m^2 (19.1–36.5), with the most common reason for removal being irregular bleeding. The majority of cases (5/6) were performed under monitored anesthesia care with local anesthetic and one case utilized regional anesthesia. All implants were surgically removed without complication. Conclusions Insertion of etonogestrel contraceptive implants deep to the biceps brachii fascia is a rare, but dangerous complication. Removal of these implants is not always successful in the office setting and referral to an upper extremity surgeon is necessary to avoid damage to delicate neurovascular structures for safe removal.

Solid implantable devices for sustained drug delivery - ScienceDirect
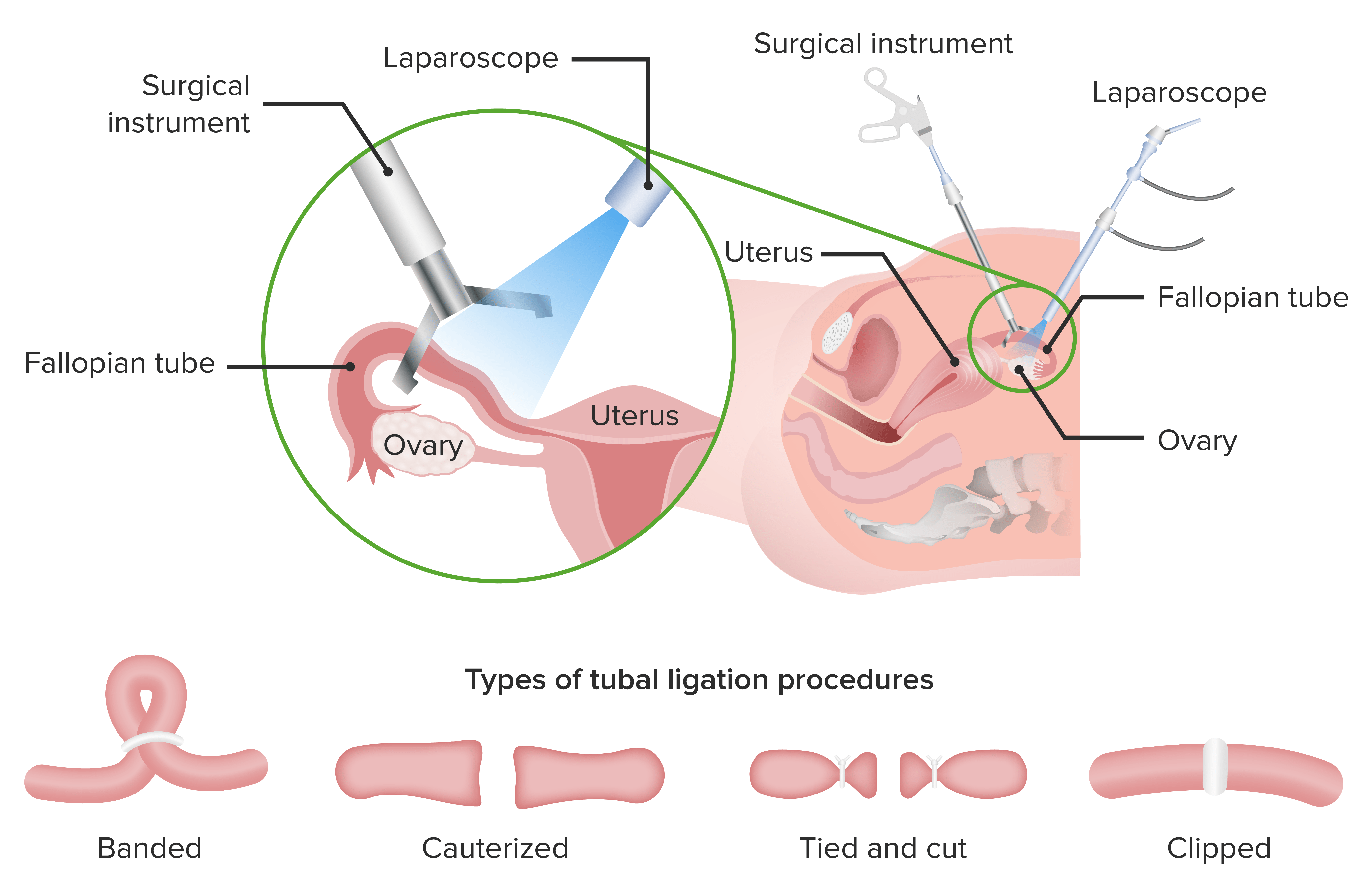
Nonhormonal Contraception Concise Medical Knowledge
Birth Control

Peripheral nerve injury with Nexplanon removal: case report and review of the literature, Contraception and Reproductive Medicine
Emergency contraception Information
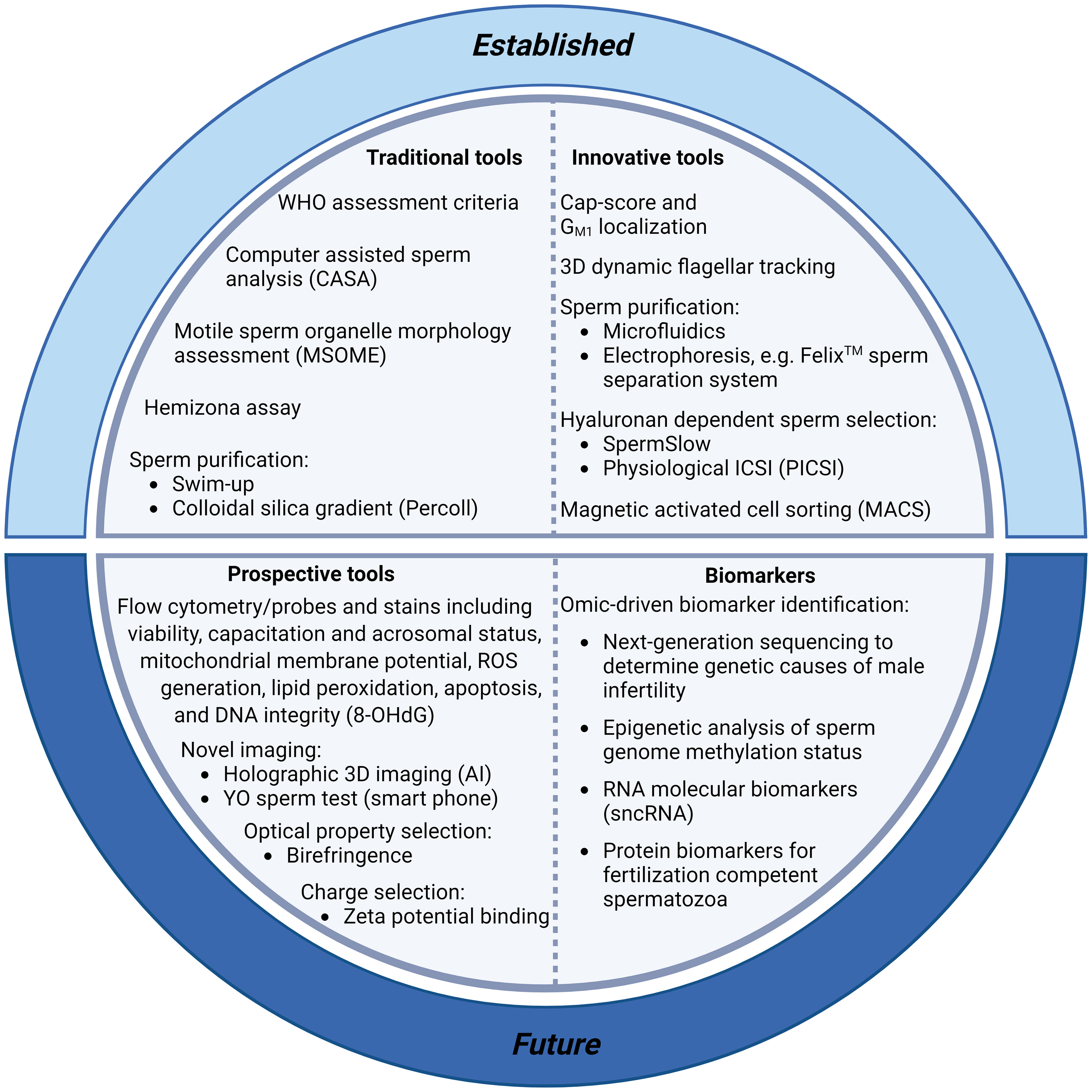
Frontiers New horizons in human sperm selection for assisted reproduction
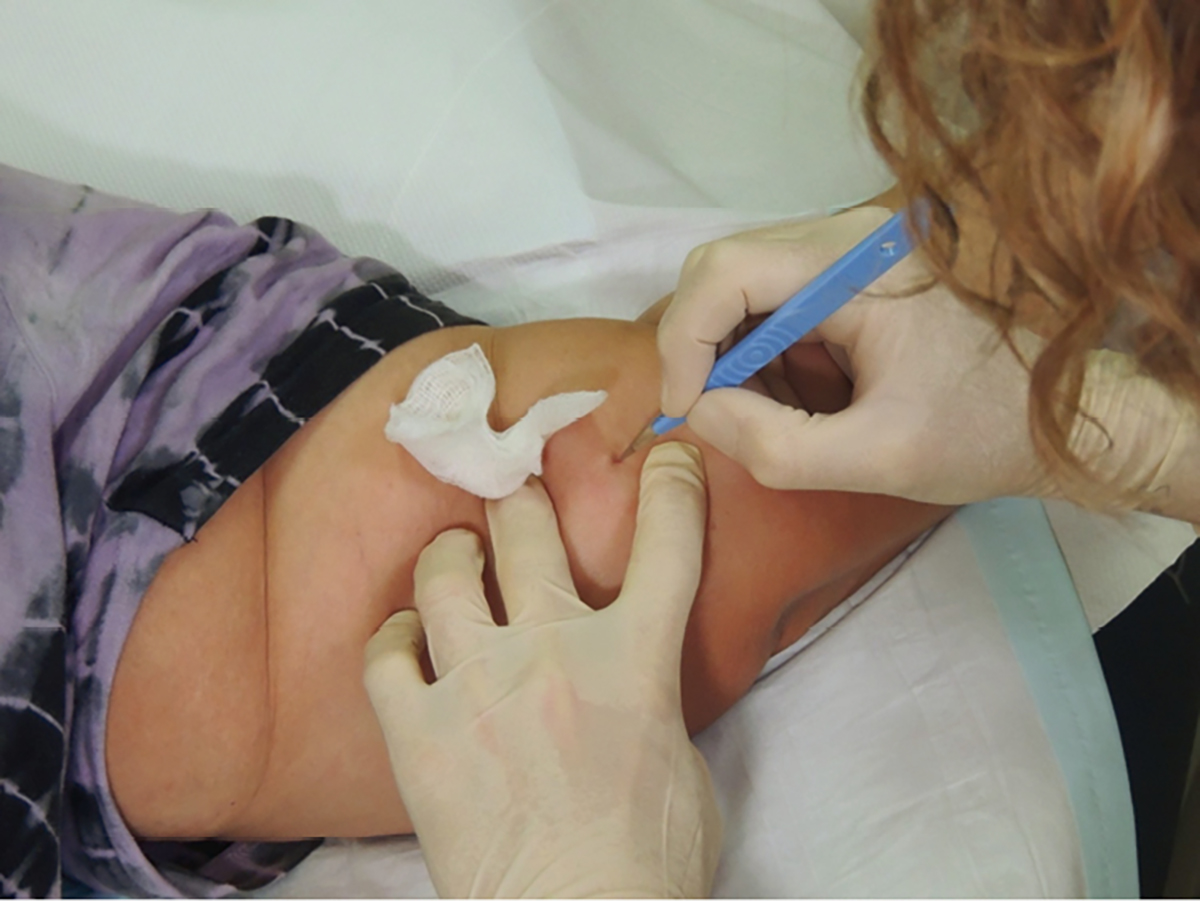
RACGP - Implanon NXT: Expert tips for best-practice insertion and removal

Difficult removal of subdermal contraceptive implants: a multidisciplinary approach involving a peripheral nerve expert. - Abstract - Europe PMC

Referral Center Experience With Nonpalpable Contraceptive Implant Removals. - Abstract - Europe PMC

PDF) Removal of non-palpable etonogestrel implants
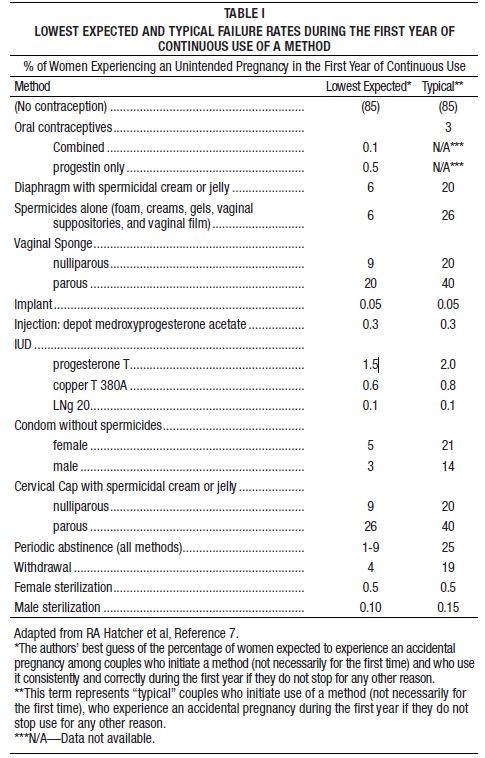
LARIN™ 1/20Norethindrone Acetate and Ethinyl Estradiol Tablets, USP
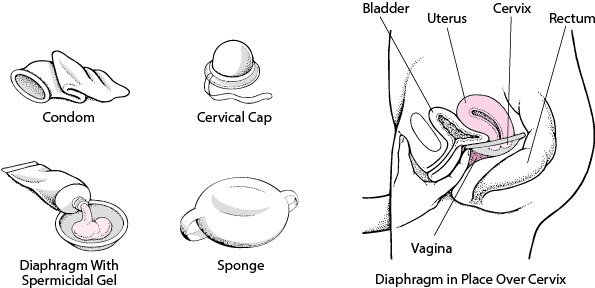
Barrier Contraceptives - Women's Health Issues - Merck Manuals Consumer Version

Article: Everything You Need to Know About the Norplant Birth Control Implant
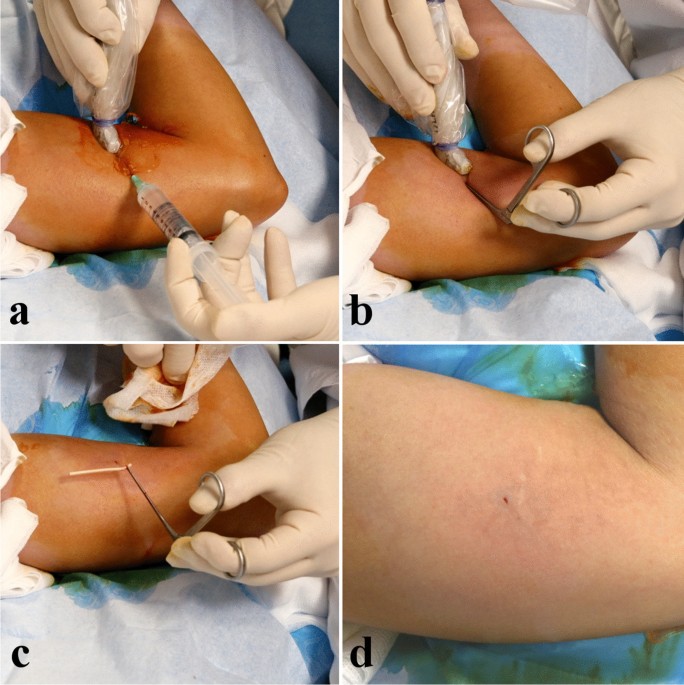
Minimally invasive removal of deep contraceptive implants under continuous ultrasound guidance is effective, quick, and safe
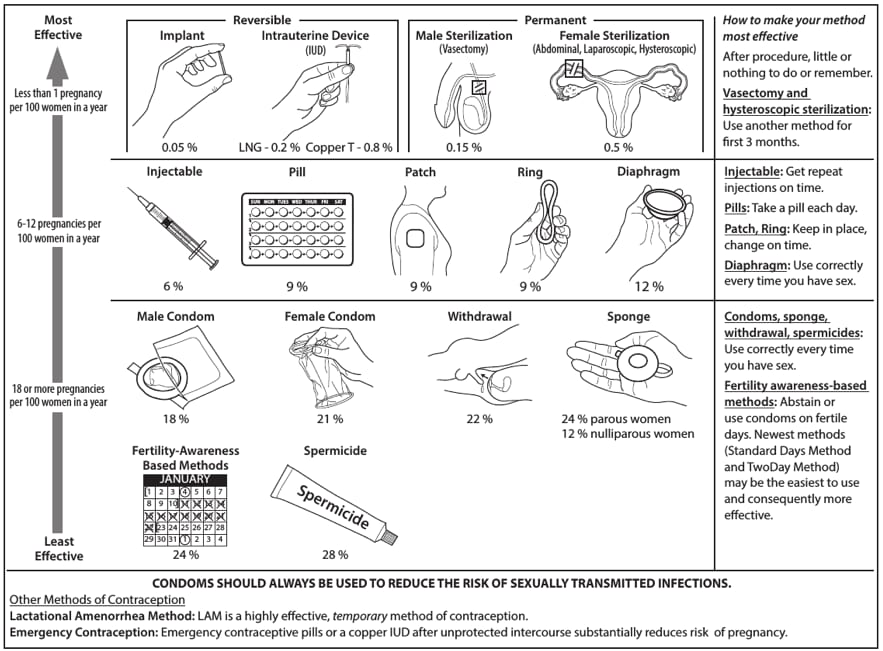
Introduction USMEC


:max_bytes(150000):strip_icc()/LULU-Chart-03282019-2539a655272444529661923139a31e79.png)
