
It is the ratio of the actual molar volume of a gas to the volume of a perfect gas under the same conditions. For a perfect gas, the value of compression factor (Z) is 1.
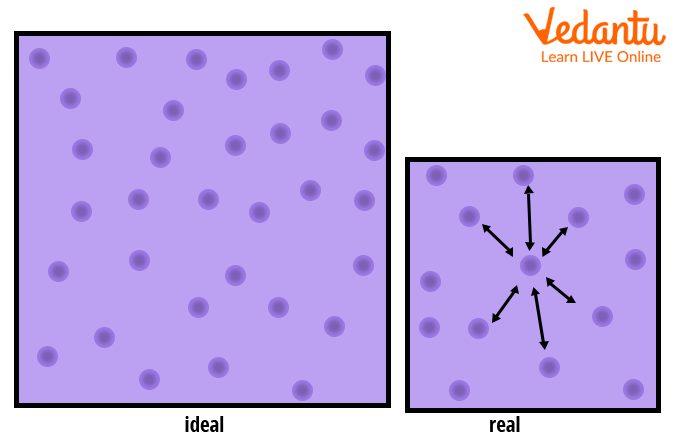
JEE - Compressibility Factor Important Concepts and Tips

Telugu] Under critical states for one mole of a gas, compressibility

The compressibility factor of a gas is defined as Z=P V / R T. The

Compressibility Chart - an overview

3.2 Real gas and compressibility factor – Introduction to

ఆదర్శ వాయు సమీకరణ – Ideal Gas equation
Compressibility Factor and Compressibility Charts

Compressibility factor - Wikipedia

Deviation Of Real Gas From Ideal Gas Behavior

Compressibility Factor (Z)

Determine Compressibility of Gases

Compressibility Factor Thermodynamics in English

Telugu] Define compression factor. What is its value for a perfect-ga







