
Share your videos with friends, family and the world

09 DEFINITION Behaviour of gases by van der Waals equation (P+*}(0-b) = RT may be written as (P+*}() =RT of PV + 9 =RT of PV=RT - For large V (at very

Calculate the critical temperature of a Van der Waals gas for which p(

Bengali] In terms of critical constants, the compressibility factor i

For a certain van der Waal's gas, critical temperature is-243^(@)C. Ma

6.3: Van der Waals and Other Gases - Physics LibreTexts

physical chemistry - Is the compressibility factor smaller or greater than 1 at low temperature and high pressure? - Chemistry Stack Exchange
If Assertion is true statement but Reason is false, then mark (3)

Compressibility factor (Z) for a van der Waals real gas at critical po
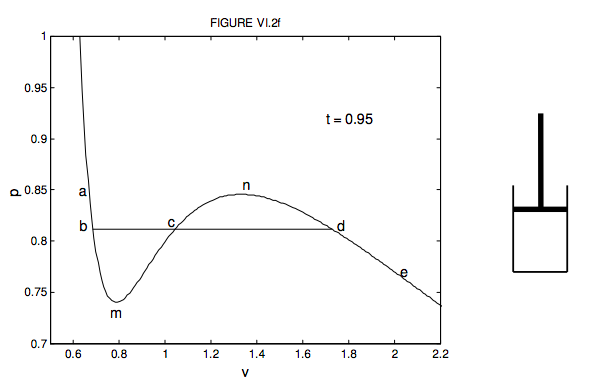
6.3: Van der Waals and Other Gases - Physics LibreTexts

Fluids, Free Full-Text
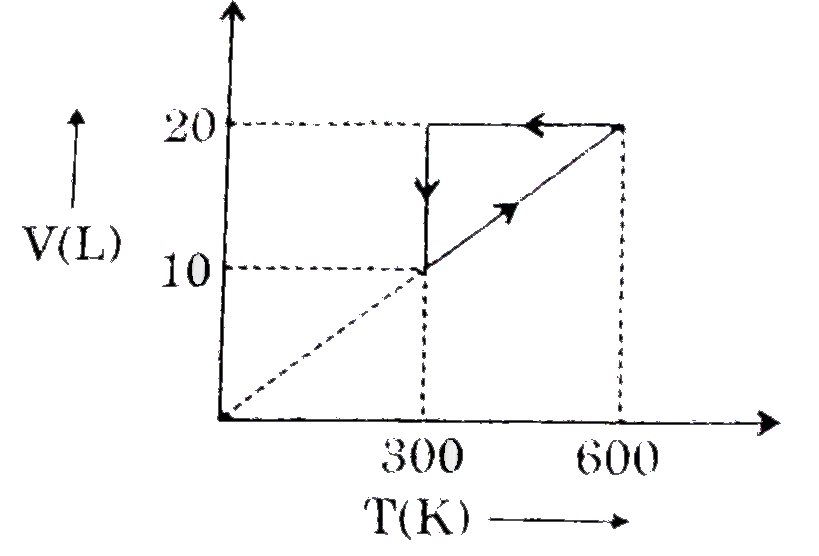
For a certain van der Waal's gas, critical temperature is-243^(@)C. Ma

Non-Ideal Gas Behavior Chemistry: Atoms First

The compressibility factor of a van der Waals gas the critical point is equal to

PDF) A Modified Form of the van der Waals Equation of State





