
Share

SOLVED: Question: Using the equation for the compressibility

Solved The van der Waals equation of state can be used to
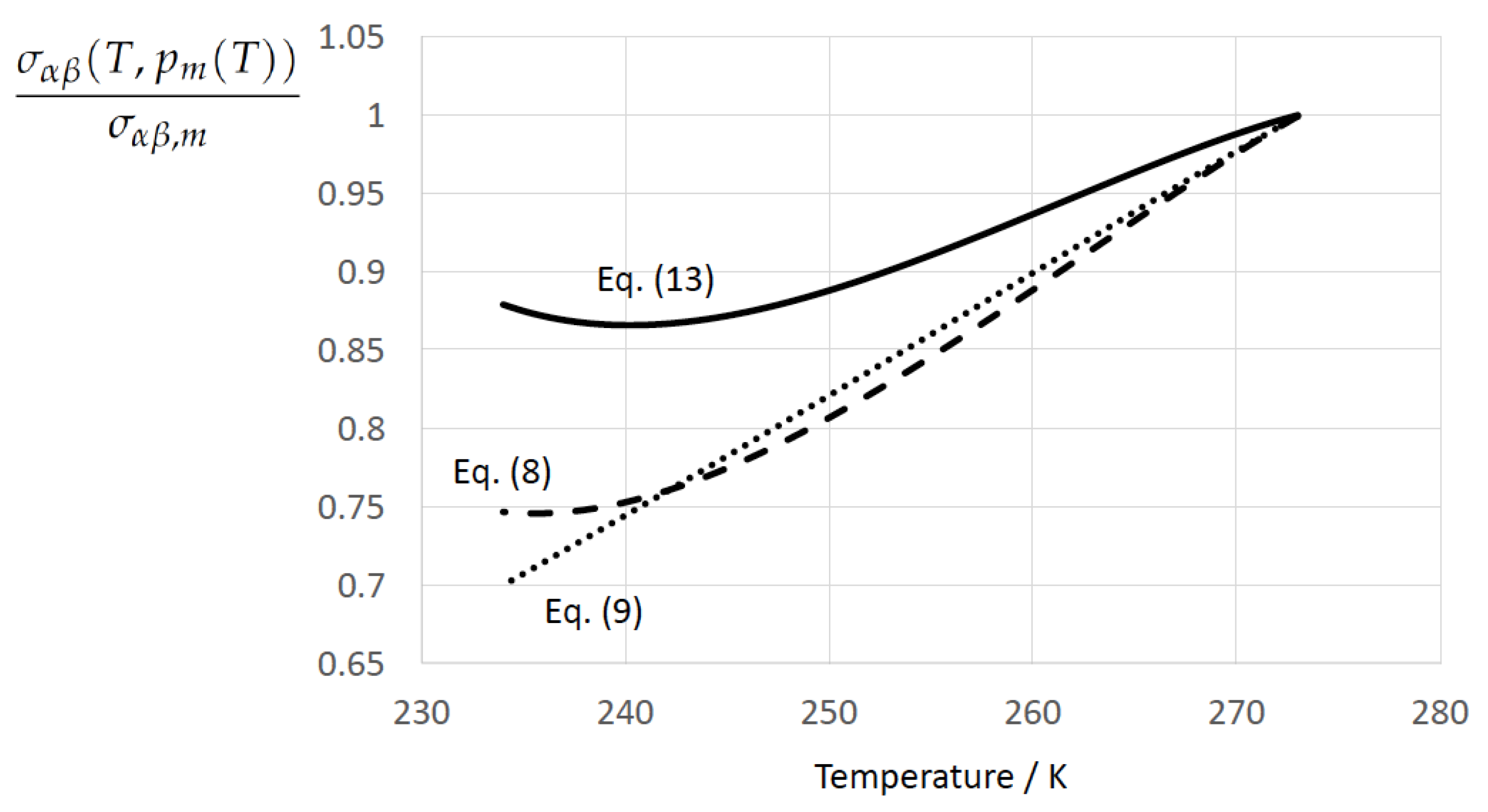
Entropy, Free Full-Text

Solved Please answer all the questions and explain how the

If `Z` is a compressibility factor, van der Waals' equation at low

Solucionario de fluidos_white

Thermo problem set no. 2
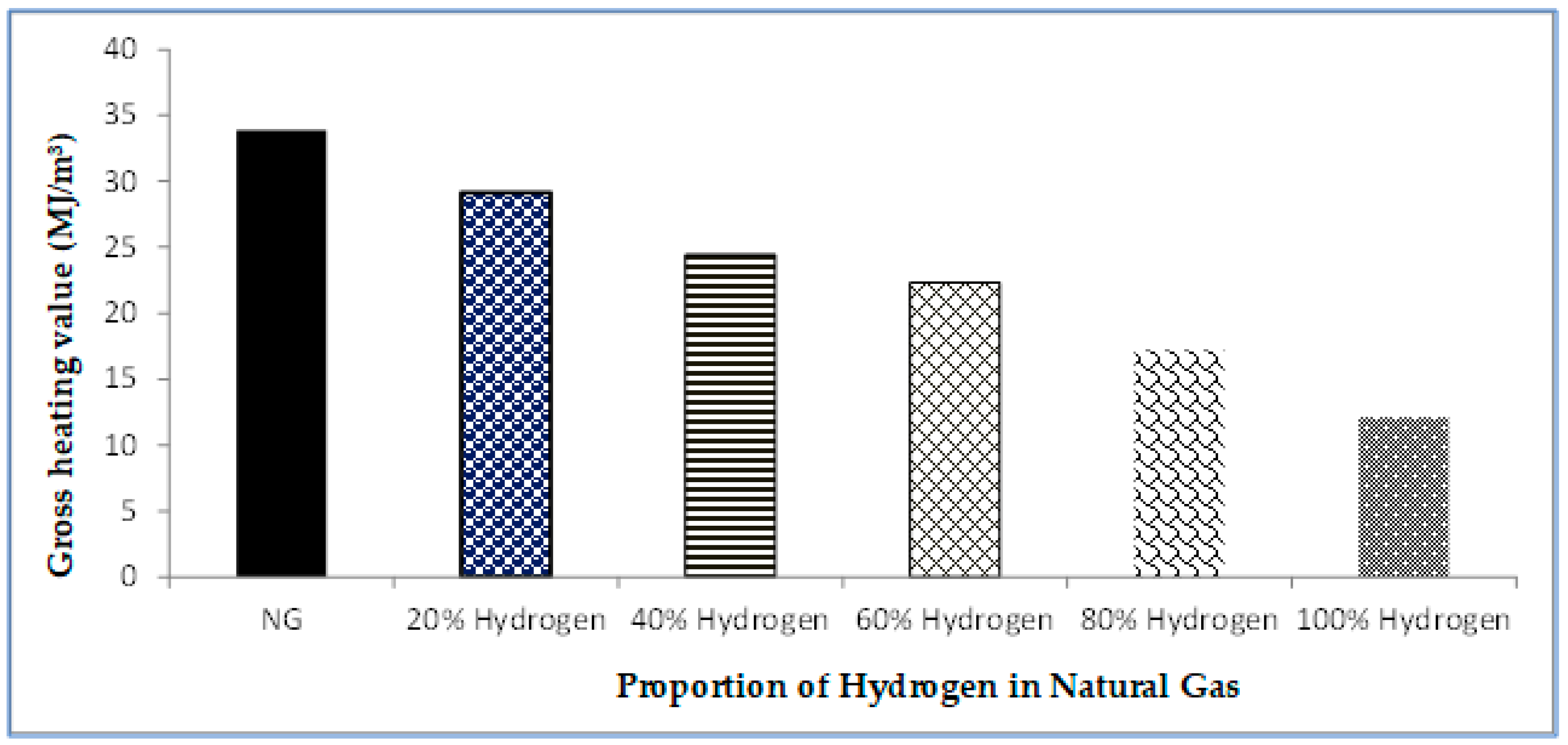
Gases, Free Full-Text

What is the compressibility factor (Z) for 0.02 mole of a van der

Solved 2. (20 points) At low pressures, the compressibility
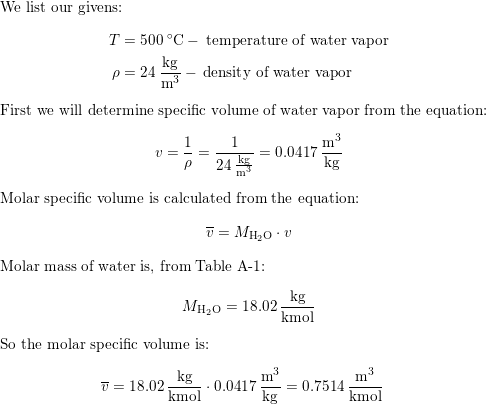
Estimate the pressure of water vapor at a temperature of $50

PDF) Chemistry 360 Problem Set 2 Solutions

SOLVED: Question: Using the equation for the compressibility
Related products

ChemE 260 Equations of State April 4, 2005 Dr. William Baratuci Senior Lecturer Chemical Engineering Department University of Washington TCD 2: E & F CB. - ppt download

The compression factor (compressibility factor) one mole of a van der Waals gas 0°C and 100 atm pressure is found to be 0.5. Assuming that the volume of a gas molecule is

Compressibility Factor Charts - Wolfram Demonstrations Project
Solved] Why is the compressibility factor less than 1 at most
You may also like



