Solution For If Z is a compressibility factor, van der Waals equation at low pressure can be written as
Video solution 1: If Z is a compressibility factor, van der Waals equation at low pressure can be written as
Video solution 2: If Z is a compressibility factor, van der Waals equation at low pressure can be written as
Video solution 3: If Z is a compressibility factor, van der Waals equation at low pressure can be written as
Video solution 4: If Z is a compressibility factor, van der Waals equation at low pressure can be written as

012 IfZ is a compressibility factor, van der Waals equation low
If Z is a compressibility factor, van der Waals equation at low

If `Z` is a compressibility factor, van der Waals' equation at low

What is the major factor that accounts for most of the difference
⏩SOLVED:If Z is a compressibility factor, van der Waals equation
If Z is a compressibility factor, van der Waals equation at low

If Z is a compressibility factor van der Waals equation at low

If Z is a compressibility factor, van der Waals' equation at low
The compression factor (compressibility factor) for one mole of a

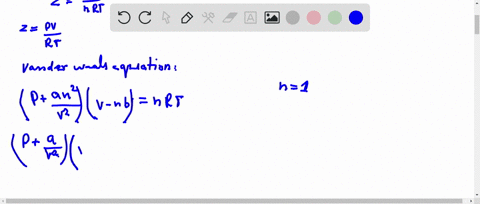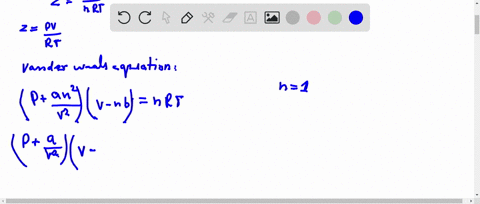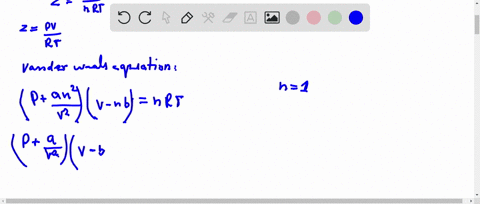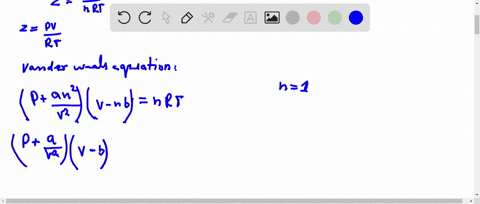
At low pressure, Van der Waal's equation is reduced to [P+dfrac{a

If Z is a compressibility factor van der Waals equation at low
The compression factor (compressibility factor) for one mole of a

Real gases 1.4 Molecular interactions 1.5 The van de Waals
If Z is a compressibility factor van der Waals equation at low

Solved The van der Waals equation of state can be used to