
Click here:point_up_2:to get an answer to your question :writing_hand:what is the mass of glucose required to produce 44g of co2 on complete
Click here👆to get an answer to your question ✍️ What is the mass of glucose required to produce 44g of C-O-2- on complete combustion-30g45g60g22g
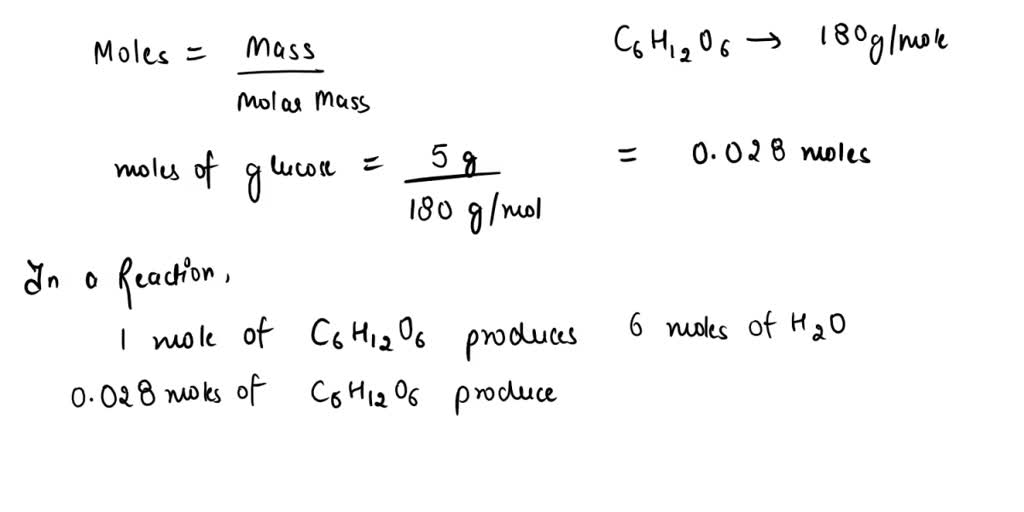
SOLVED: How many grams of water are produced in the combustion of 5.00g of glucose, C6H12O6? C6H12O6(s) + 6 O2(g) â†' 6 CO2(g) + 6 H2O(l)
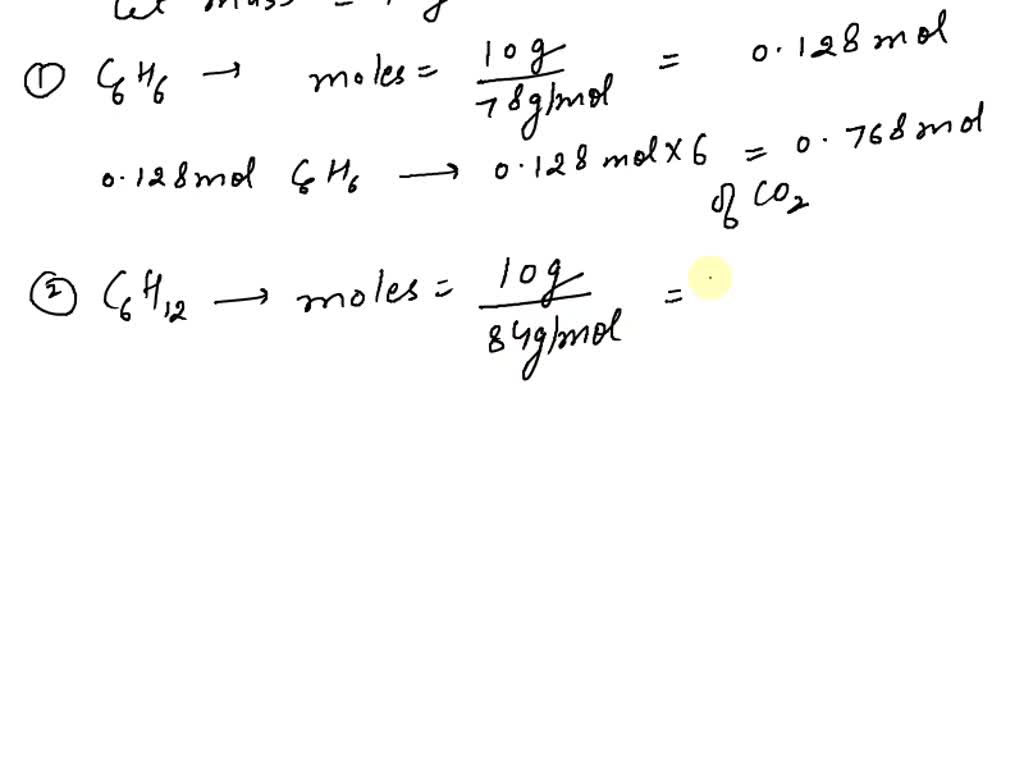
SOLVED: If equal masses of the following compounds undergo complete combustion, which will yield the greatest mass of CO2? 1. Benzene, C6H6 2. Cyclohexane, C6H12 3. Glucose, C6H12O6 4. Methane, CH4
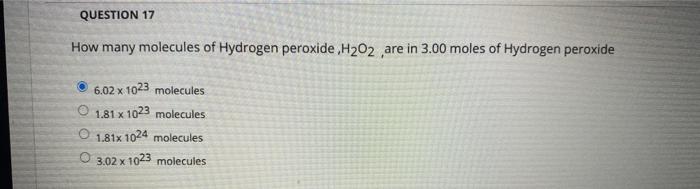
Solved QUESTION 15 5 when 2.43 grams of Mg reacts with
Mass of glucose required to produce 44g of co2 on complete oxidation?

Solved 3. Consider the combustion of the sugar sucrose

SOLVED: If equal masses of the following compounds undergo complete combustion, which will yield the greatest mass of CO2? 1. Benzene, C6H6 2. Cyclohexane, C6H12 3. Glucose, C6H12O6 4. Methane, CH4
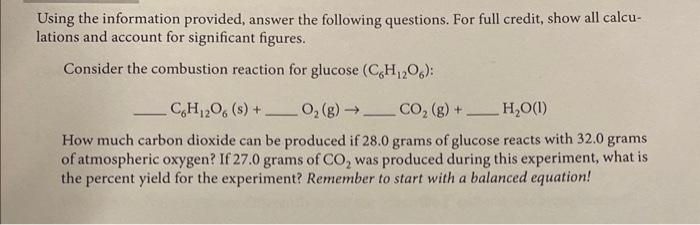
Solved Using the information provided, answer the following
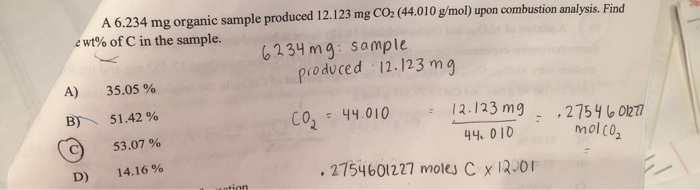
Solved A 6.234 mg organic sample produced 12.123 mg CO2

what is the mass of glucose required to produce 44grams of CO2 on complete combustion - 10ijyqww
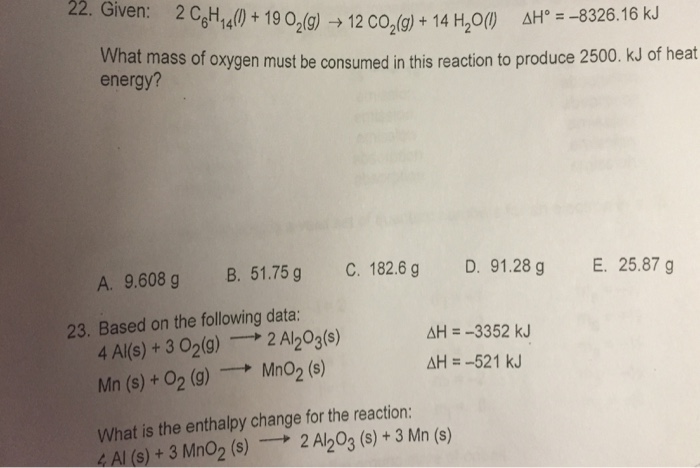
Solved 22 Given: 2 C, H140+19 C2 2 G c6H14() + 1902(g) → 12

Determine the mass of CO2 produced by burning enough of each fuel
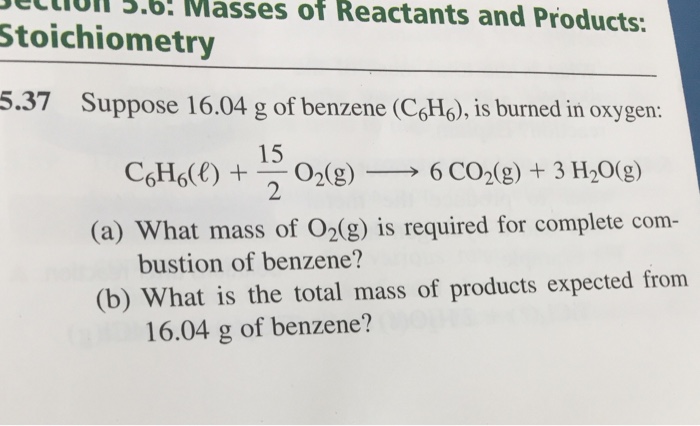
Solved Suppose 16.04 g of benzene (C_6 H_6), is burned in

Solved Consider the combustion of 5.000 g of glucose

:quality(80)/anatofee/catalog/7653-01-3944-g.jpg)





